Pool Chemistry for Beginners: With 5 Super Helpful Cheat Sheets!
Table of Contents
Pool Chemistry for Beginners: Getting Started!
Knowledge of Pool Chemistry for Beginners is absolutely essential for new pool owners! Believe it or not, the difference between crystal-clear pool waters and a murky unhygienic nightmare may be just a slight imbalance in pool chemistry.
This easy-to-understand Pool Chemistry for Beginners will get you started and will be a helpful guide for years to come.
This post is a definitive guide that empowers you with the knowledge and expertise to understand concepts of pool sanitization, pH, alkalinity and calcium hardness levels, etc.
But why not equip yourself with an interactive tool that is like a Swiss knife for pool water chemistry?
Pool chemistry is all about knowing the right level of all the chemicals that should be in your pool, testing to determine the actual levels and then adding chemicals or taking other steps to get the actual levels to the desired or ideal levels.
All pool chemistry parameters, except for pH are expressed as “parts per million” (ppm).
Testing the Pool Water

Testing pool water to determine current chemical levels is the first step in pool chemistry balancing for both beginners and pros. Get a pool water test kit from a local pool supply store or online.
There are three options available.
- Test Strips
- Test Kits using chemicals
- Electronic Test Kits
Test strips are sufficient for beginners; they are inexpensive and easy to use. Simply dip the strip in the pool water to get a reading. However, you will soon want to graduate to Test Kits using Chemicals.
The article The Best Pool Testing Kits from Pool Research is a good resource and worth reading too!
Before the First Fill
The water you use to fill your pool for the first time isn’t completely pure and free of chemicals; both tap water and groundwater contain various chemicals.
It’s best to take a sample to your local pool dealer for testing. They have advanced testing equipment and will provide a detailed report on the chemical levels.
If you’re unsure, ask them to set up your pool water to ensure perfect chemistry for the swim season. Alternatively, you can buy the chemicals and add them yourself.
Know your pool volume. You will need this information all the time for adding the right quantity of pool chemicals. Use this link to my pool volume calculator.
All the Necessary Pool Chemicals – Summary
This pool chemistry for beginners article will guide you through the first time as well as regular pool chemistry balancing. So, here’s what you need to know about the chemicals you’ll need to add to your pool for the first time.
All the chemicals that you need can be divided into 2 broad groups, based on their function, as under:
ABSOLUTE “MUST HAVES”
- 1. Sanitizer (Chlorine)
- 2. Shock (Concentrated Chlorine)
- 3. Stabilizer (Cyanuric Acid)
- 4. pH Related (Soda Ash, Muriatic Acid, Sodium Bisulfate)
- 5. Alkalinity Related (Baking Soda, Muriatic Acid, Sodium Bisulfate)
- 6. Calcium Hardness (Calcium Chloride)
IMPORTANT (“GOOD TO HAVE”)
- 7. Algaecide
- 8. Clarifier
- 9. Flocculant
- 10. Sequestering Agents
This Pool Chemistry for Beginners Guide gives easy to understand information on each.
(Don’t worry if you have little or even no knowledge of chemistry. You will know, all that you need to know, after reading this article)
Click on the image below to reach the page with all the Necessary Pool Chemicals.
All the Absolute “Must Have” Pool Chemicals – Detailed
1. Sanitizer (Chlorine)
Pool chemicals must keep the pool sanitized & safe for swimming, clear & looking good and should not corrode or damage the pool and its equipment.
Without doubt, the most critical chemical for your pool is the “Sanitizer”. Your pool will have a a continuous influx of contaminants, bacteria, virus, algae and other microorganisms. Many of them are harmful.
A pool sanitizer is the chemical that fights and neutralizes the pathogens. There are a few choices, but chlorine is by far the most common.
Other options include bromine and biguanide. As a beginner to pool chemistry, using chlorine as a sanitizer for your pool is a safe choice. This is what you need to know:
What is Chlorine?
Chlorine is an element and is a gas in its natural state. However, that’s not how you use it in your pool. Instead you use a compound of chlorine which is either available as a powder or a solution in water.
The 4 most commonly used types of chlorine for pool are:
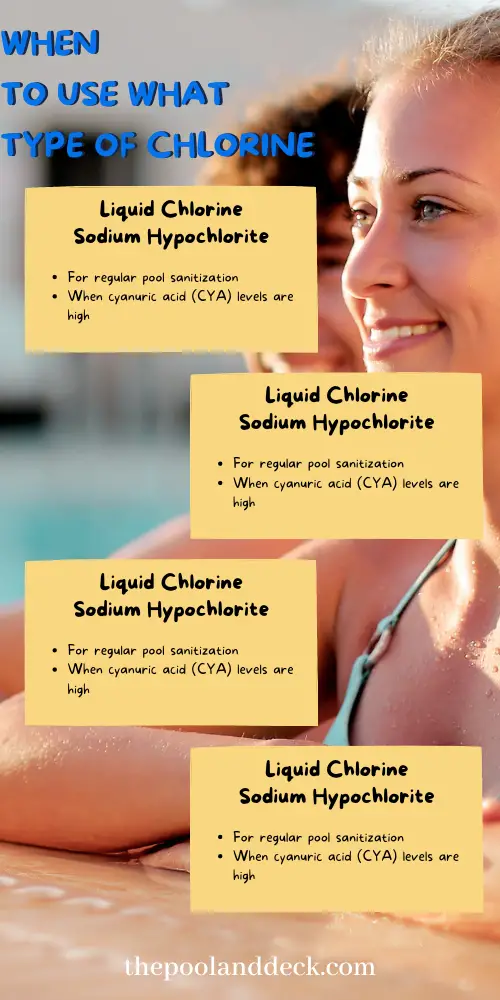
Sodium Hypochlorite (Liquid Chlorine) is the salt of Sodium with Hypochlorous Acid. It is sold as a 10-12.5% solution of Sodium Hypochlorite in water, hence the name Liquid Chlorine. It is just a stronger version of the household Bleach, which is a 6% solution. It can be used for regular pool sanitization in case Cyanuric Acid levels are high.
Calcium Hypochlorite (Cal Hypo) is the salt of Calcium with Hypochlorous Acid. It is usually sold as a granular powder but is also available as “slow dissolve” tablets. Cal Hypo, for pool use, typically has 65-70% Available Chlorine (AC) and is generally used as a shock, rather than a regular sanitizer .
Dichlor (Dichloro-S-Triazinetrione) is usually sold as a granular powder but can also come as tablets. The powder is most commonly used for shocking a pool. More on this when we talk about “shock” and “stabilizer”.
Trichlor (Trichloro-S-Triazinetrione) is most commonly sold as 1” or 3” tablets for sanitizing home swimming pools. The tablets can be inserted in the skimmer or in floaters. This is very convenient as you can load the tablets for a week at a time. Trichlor is also the richest source of chlorine as it has 90% available chlorine.
So When to use What Type of Chlorine? Here is the Cheat Sheet!
Pool Chemistry Cheat Sheet # 1: When to Use What Type of Chlorine
How does Chlorine Sanitize?
The four Pool Chlorine Types are essentially chlorine based compounds that react with the pool water to form a weak acid called Hypochlorous Acid (HOCL).
The Hypochlorous Acid (HOCL) sanitizes & disinfects by killing the viruses, germs, bacteria & algae. The acid penetrates the cell walls of the pathogens and makes the cell’s protein backbone dysfunctional.
In addition Chlorine is also an “oxidizer”. Which means that chlorine can break down and destroy non-biological contaminants too!
The chlorine in your pool that still has the power to kill bacteria is called “Free Chlorine (FC)”. The chlorine that has already been spent is called “Combined Chlorine (CC)”. Together they are known as “Total Chlorine (TC)”. It is important to know this as testing kits only give TC and FC levels.
Combined Chlorine, which mostly consists of Chloramines, is not a good thing to have in your pool. By the way, the distinct chlorine smell that you get from your pool is actually from chloramine. You need to lower Combined Chlorine levels.
What are Ideal Chlorine Levels?
In the ideal situation, Combined Chlorine levels should be 0 ppm and you should not let it be higher than 0.5 ppm.
The range for Free Chlorine is 1 – 4 ppm. It is great if you can keep it in the 2 – 3 ppm range. Higher than 5 ppm will make your pool water acidic and cause skin & eye irritation and corrosion. Lower than 1 ppm will result in a poorly sanitized pool that can result in waterborne diseases and “algae blooms”.
For more detailed information you can check out my earlier blog post Complete Guide To Pool Chlorine Types: Which One Is The Best?
2. Shock (Hyper Chlorination)
What is Shock?
In terms of chemical composition regular chlorine and “shock” are pretty much the same thing. The difference is in the concentration, rate of release and the objective.
While regular chlorine is released in the pool at a slow steady pace to keep the FC level within 1 – 4 ppm, shock is hyper chlorination. Pool shock is intended to spike up the FC level of the pool to around 30 ppm.
Out of the 4 types of chlorine mentioned above, the two that are commonly used as shock are Cal Hypo and Dichlor. Both are available as granular powder. They can be sprinkled on the pool surface but are usually pre-dissolved in water to facilitate quick dispersion.
Powder shock such as Cal Hypo and Dichlor are very effective as they have a very high %age of active chlorine. Cal Hypo has 65% – 70% and Dichlor has 56%.
Why do you Need to Shock your Pool?
Shock or hyper chlorination spikes up the chlorine levels for a short period of time, when the pool will not be in use, to eliminate excessive contaminants, harmful pathogens and algae outbreaks. Think of it like a “deep clean”.
When Should You Shock Your Pool?
Shocking your pool will go a long way in keeping your pool free of contaminants, pathogens and algae. I recommend that you shock your pool
- At least once a week during the pool season.
- After heavy pool usage such as after a pool party. Swimmers are the source of non-living organic matter in the water, such as sweat, sunscreen, and other body oils & fluids.
- After heavy rainfall, high winds or other environmental factors that may have introduced contaminants into the pool water.
- If you notice a strong chlorine odor, unusual color or signs of algae growth.
- On opening the pool in spring and before closing it for winter.
The best time to shock a pool is in the evening. Keep in mind that the pool can not be used during shock as the chlorine levels are dangerously high.
3. Stabilizer (Cyanuric Acid)
Why do you Need a Stabilizer?
Chlorine is a great pool sanitizer but Free Chlorine (FC) breaks down very rapidly under UV rays of the sun. UV rays can reduce the half life of Free Chlorine (FC) to as low as 45 minutes.
So if you start off with 4 ppm of free chlorine, you are down to just 2 ppm in 45 minutes and only 1 ppm in 90 minutes.
Clearly free chlorine needs some protection, when bombarded by UV rays. It needs sunscreen. Cyanuric Acid (CYA) is that sunscreen for chlorine and is referred to as a “stabilizer” in pool chemistry.
Stabilized & Unstabilized Chlorine
The 4 types of pool chlorine mentioned above are stabilized or unstabilized as under:
| STABILIZED | UN-STABILIZED |
| Dichlor (Dichloro-S-Triazinetrione) | Sodium Hypochlorite (Liquid Chlorine) |
| Trichlor (Trichloro-S-Triazinetrione) | Calcium Hypochlorite (Cal Hypo) |
Ideal CYA Level
The ideal range for cyanuric acid (CYA) is 30-50 ppm to ensure optimal pool health and sanitation.
High CYA Levels
But, if cyanuric acid levels get too high, it can actually have the opposite effect. High levels of cyanuric acid can lead to a condition known as “chlorine lock“. Free chlorine becomes bound to the cyanuric acid and is unable to effectively sanitize the pool.
High cyanuric acid levels can result in cloudy water and increased risk of algae growth. I have a separate post on how to deal with high cyanuric acid in your pool.
Low CYA Levels
Very low cyanuric acid level, on the other hand, results in quick depletion of Free Chlorine (FC) due to the effects of UV rays. In this case there is never enough chlorine available to effectively sanitize the pool.
Low cyanuric acid levels lead to increased chlorine demand. You will be adding a lot more chlorine for the same level of sanitation levels.
4. pH & Alkalinity
I decided to club the discussion on pool pH & Alkalinity under the same heading as the two are so intertwined. To a beginner pH & alkalinity seem to mean the same thing. They are closely related but they are not the same thing.
pH is a measure of the acidity or the alkalinity of the pool water. Total Alkalinity (TA) is a measure of the amount of alkaline particles dissolved in the water. The alkalinity of water is its ability to neutralize acids. Alkalinity therefore acts, and is known as a pH buffer!
How is pH Measured?
A logarithmic scale from 0 to 14 is used to measure pH. Distilled water at a pH of 7 is considered neutral. A pH level below 7 indicates that your pool water is acidic, while a pH level above 7 indicates that your water is alkaline.
Note that the scale is logarithmic. So water with a pH of 6 is not just a bit more acidic than water with a pH of 7, considered neutral. It is 10X more acidic!
Maintaining proper pH level is crucial. In case pH is low and your pool water is too acidic, it can cause eye and skin irritation, damage your pool equipment, and even corrode your pool walls and floor.
Use pH Up (also sold as pH Increaser or pH Plus). The active ingredient in all of them is sodium carbonate (soda ash).
On the other hand, if pH is high and your pool water is too alkaline, it can cause calcium deposits on your pool surfaces, reduce the effectiveness of chlorine, and make your water cloudy.
Use pH Down (also sold as pH Decreaser or pH Minus). The active ingredient in all of them is either muriatic acid or sodium bisulfate (dry acid).
How is Total Alkalinity (TA) Measured?
Total Alkalinity is a measure of the ability of pool water to resist changes in pH. It is a measure of the quantity of alkaline particles in the water and is measured in parts per million (ppm).
Total Alkalinity is a buffer that stabilizes pH levels and prevents rapid changes in it.
Low Total Alkalinity can cause the pH to fluctuate rapidly, which can damage your pool equipment and surfaces. On the other hand, if your alkalinity is too high, it can make it difficult to adjust the pH level, which can cause cloudy water and reduce the effectiveness of chlorine.
Use sodium bicarbonate (baking soda) to get alkalinity up and either muriatic acid or sodium bisulfate (dry acid) to get alkalinity down.
Ideal pH & Alkalinity Levels
The acceptable pH range of pool water is 7.2 to 7.8. You should try to keep it within 7.4 to 7.6 for the best swim experience.
Alkalinity level should never be allowed to go below 80 ppm as it will cause the pool pH to fluctuate wildly. On the other hand it should not be allowed to go above 150 ppm. The ideal range is 80 – 120 ppm.
4. Calcium Hardness
Calcium Hardness (CH) is a measure of the amount of dissolved calcium ions in the pool water. It is an important parameter in pool chemistry. Water with low calcium hardness is referred to as “soft water” and that with high calcium hardness as “hard water”.
High calcium hardness will result in calcium deposits on pool plumbing, pumps and filters leading to corrosion and reduced lifespan. It will also cause scaling of pool walls & bottom making them rough.
A rough pool is more likely to promote algae growth. High calcium hardness will also cause the pool water to appear cloudy.
Ideal Calcium Hardness (CH) Levels
Calcium Hardness (CH) levels between 150 and 400 ppm are considered acceptable. However, the ideal range for calcium hardness is 200 – 300 ppm. You should not, in any case, let it go above 400 ppm.
You can increase Calcium Hardness (CH) by adding Calcium Chloride. The most effective way to reduce Calcium Hardness (CH) level is by partial draining and replacing with fresh water. The process must be really gradual!
Pool Chemistry Cheat Sheet # 2: Acceptable & Ideal Range
The infographic below gives the acceptable and ideal range for the 5 key pool chemistry parameters.

Other Important & “Good to Have” Pool Chemicals
The chemicals that I have talked about so far, will get you through almost all pool chemistry related issues. However, there are 4 more that are worth keeping in stock and using when the need arises.
Algaecide
Algae growth is a fairly common occurrence in a pool, especially during the warmer months. Now, to be fair, algaecide does not really kill algae, but it does help prevent algae growth. (Only chlorine does the killing).
Algaecide weakens the algae making it easier for chlorine to kill them. Algaecide acts by disrupting energy transport, cell division, or the cell membranes of the algae.
Using algaecide, will however, have your back covered should the chlorine levels dip or there is a deluge of algae spores. Use algaecide at following times:
- After shocking the pool and once FC drops below 5 ppm
- Before & after a thunderstorm
- Before pool closure
- Just after pool opening
- If your pool appears cloudy
Clarifier
Sometimes, despite regular maintenance, your pool water can still appear cloudy or hazy. This is the presence of particles suspended in the pool water that are too small to get filtered out.
This is where a clarifier comes in. Clarifier helps to gather together the tiny particles and form larger ones that can be trapped by the pool filter. Add it to the pool water while the filter is running to turn your cloudy pool to crystal clear.
Flocculant
Sometimes, especially after a storm or chlorine shock, the pool may be so cloudy that you can’t even see the pool bottom. Basically the quantity of microscopic solid particles or dead algae from the shock is very high.
Do not use a pool clarifier in such a situation as the pool filter will keep getting clogged, every now and then. Use a pool flocculant instead!
The pool flocculant will coalesce the microscopic solid particles and make them sink to the pool floor. You should then use a pool vacuum to suck up the muck at the pool bottom and discharge it into the yard or a drain.
Both clarifiers and flocculants help turn a cloudy pool into a crystal clear one. If you must use both, use the flocculant first.
Metal Sequestering Agents
Metal sequestering agents are used to prevent and remove metal stains from pool surfaces. Metal stains are caused by high levels of minerals such as copper, iron or manganese in the water.
Metal sequestering agents work by binding to the metal ions in the water and preventing them from reacting with other chemicals, thus preventing staining and corrosion.
Pool Chemistry Cheat Sheet #3: All the Pool Chemicals you Need
The infographic below is a list of all the pool chemicals you need!

Should Pool Chemicals be Added in any Sequence?
A very important point about pool chemistry that beginners must always keep in mind is that the various pool chemicals do not function independent of each other. They influence the action of each other in many cases.
Therefore the sequence in which the chemicals are added to the pool affects the final outcome.
The Recommended Sequence
The sequence in which the chemicals must be added depends on the actual levels from the pool water test. If you are planning to shock your pool then I recommend the following sequence:
Alkalinity Adjustment:
Always adjust the alkalinity level of your pool water first. Check out my free alkalinity pool calculator.
Once alkalinity is adjusted to above 80 ppm (preferably above 100 ppm) it will become a pH buffer. It will be a lot easier to adjust pH after that.
Adjust the pool pH:
Adjust pH to between 7.2 and 7.4 before shocking the pool. Shock (chlorine) is more effective when pH is at the lower end. Of course you will need to readjust all parameters including pH once the shock process is complete.
Calcium Hardness:
Calcium Hardness (CH) needs to be adjusted upwards only if it is below 175 – 200 ppm. If at this level, just let it be. Cal Hypo Shock will naturally increase calcium hardness.
Shock:
The choice of shock depends on the Cyanuric Acid (CYA) level of the pool. If it is lower than 30 ppm you may use Dichlor to shock your pool. However, at higher levels you must use Cal Hypo shock as it is un-stabilized and will not add to the CYA level.
After the shock process is complete, retest the water. You may need to once again fine tune the pH and alkalinity levels.
Flocculant & Clarifier:
It is not unusual for your pool to be quite cloudy after shock. This is the neutralized contaminants, pathogens and algae floating around.
Add the flocculant first. Brush the sides and step to push the debris to the pool floor. Vacuum out the debris. Follow up with a clarifier while running the pool pump and filter. You will get a crystal clear pool.
Algaecide:
Addition of algaecide at this stage will help fortify chlorine for its war against algae in the future.
The level of certain chemicals and minerals such as cyanuric acid (CYA) or calcium keep increasing over time. The most effective way of reducing their level is by diluting the pool water.
Do this by draining a little of the pool and filling it up with fresh water. Be extremely careful that you execute this process in small incremental steps. An inground pool can pop due to hydrostatic pressure if it is drained too much.
NOTE: If you are required to dilute a pool chemical such as Cal Hypo powder, always add the chemical to water and not the other way around.
It’s important to follow the correct sequence of adding pool chemicals to ensure that they work effectively and don’t interfere with one another. Adding chemicals out of order can cause imbalances in the water chemistry which may be quite difficult to set right.
Pool Chemistry Cheat Sheet #4: The Sequence of Adding Pool Chemicals
The infographic below lists out the correct sequence of adding pool chemicals.

Troubleshooting Common Pool Chemistry Related Problems
A pool will, every now and then throw up some problems. As a beginner they may stump you but usually there is a solution. Below is a list of common problems and recommended solutions.
Algae Growth
Algae Growth: This is a very common problem faced by pool owners, especially beginners. Algae growth can occur due to improper pH levels, inadequate sanitizer levels, or poor circulation. To prevent algae growth, I recommend maintaining proper pH levels (7.2-7.8), shocking the pool with a chlorine-based shock, and brushing the walls and floors regularly to remove any algae spores.
Chlorine Loss
Chlorine Loss: Chlorine loss can occur due to high temperatures, heavy rainfall, and high bather loads. To prevent chlorine loss, test the pool’s chlorine levels regularly and increase chlorine dosage. It is better to use Trichlor which is a stabilized chlorine and will withstand UV rays better.
Combined Chlorine
Combined Chlorine: Combined chlorine occurs when chlorine combines with ammonia or other organic compounds in the water. The end product is Chloramines. It can cause eye & skin irritation and give the water a strong chlorine odor. To lower combined chlorine, shock the pool.
Cyanuric Acid Buildup
Cyanuric Acid Buildup: Cyanuric acid is a stabilizer that is commonly used to protect chlorine from UV rays. However, excessive buildup of cyanuric acid can cause chlorine to become less effective and lead to cloudy water. To prevent cyanuric acid buildup, switch from stabilized to non-stabilized chlorine. Eventually you may have to consider diluting the pool water by partial drainage and refill with fresh water.
pH Imbalance
pH Imbalance: pH imbalance can cause skin and eye irritation, corrosion of pool equipment, and cloudiness in the water. To maintain the proper pH balance, test the pool’s pH levels regularly and adjust them using pH Up or pH Down as needed.
Total Alkalinity
Total Alkalinity: Total alkalinity is a measure of the pool’s ability to resist changes in pH levels. Low total alkalinity can cause pH levels to fluctuate rapidly, leading to corrosion of pool equipment and skin & eye irritation. Test the pool’s total alkalinity levels regularly and adjust them using alkalinity increaser or decreaser as needed.
Hard Water
Hard Water: Hard water (high calcium hardness) can cause scaling on the pool’s walls and equipment, as well as cloudiness in the water. To prevent hard water, use a water softener or a sequestering agent that will prevent minerals from bonding together and causing scaling. If calcium hardness levels are over 400 ppm consider diluting the pool water by partial drainage and refill.
Phosphates
Phosphates: Phosphates are nutrients that can enter the pool water from rain, debris, or other sources. They can contribute to algae growth and cloudy water. To eliminate phosphates, use a phosphate remover product and maintain proper filtration and circulation.
Pool Chemistry Cheat Sheet #5: Troubleshooting Common Problems
The infographic below is a quick checklist of common pool chemistry problems & recommended solutions.

I hope that this “Pool Chemistry for Beginners” has been useful! Thanks for reading. If you like it, bookmark the article or take a screenshot of the cheat sheets for ready reference.
Click on the image below to reach the page with all the Necessary Pool Chemicals.
Thank you very much for reading the post. I do hope you found it informative and helpful.








